Home Creme de Vape Blog
Creme de Vape Blog
SAY NO to the proposed ban on flavoured e-liquids
SAY NO to the proposed ban on flavoured e-liquids for e-cigarettes.
Vaping has been a lifeline for many, helping them break free from deadly tobacco. Let's not turn back the clock on progress.
Sign this petition to speak up for your access to flavoured e-liquids. https://petition.parliament.uk/petitions/656683 and be sure to follow this up with an email to your MP. You can contact your MP here - www.writetothem.com
If their plans go ahead and all flavoured e-liquids are banned, we'll all have to say goodbye to our favourite fruity, dessert, and sweet flavours. The only flavour available will be tobacco.
Your choices have already been impacted by ill-thought-out, fear-based legislation. This proposed ban on flavours is not going to help with the teen vaping issue but it will seriously impact adults who use flavoured vapes responsibility to stay off combustible tobacco.
Make your voice heard!
WE'RE MOVING - YOU'RE SAVING!

WE'RE MOVING - YOU'RE SAVING!
We are relocating our warehouse and offices this week to a new location so there will be no shipping Thursday 8th - Friday 9th and the Click & Collect service will be suspended on those days.
Check our Current Shipping Status page for updates and our Click & Collect page for our new location details.
Use the coupon code MOVE10 to receive 10% off site-wide until midnight 11th Feb 2024. Also applies to already discounted items.
We hope this discount offer makes up for any inconvenience caused by this outage.
Coupon must be added before checkout is completed and cannot be applied retrospectively.
One coupon code per order.
You may use the code as many times as you like.
Discount also applies on already reduced items.
Not valid on postage and packaging costs.
Valid until midnight on Sunday 11th Feb 2024.
Urgent Call to Action: Protect Your Vaping Choices!

We are reaching out to you today with a matter of utmost importance that affects every vaper in the UK.
The Department of Health and Social Care is currently conducting a consultation about the plans to severely restrict the flavours and the types of products you currently enjoy.
At Crème de Vape, we have been dedicated to providing you with high-end vaping equipment and supporting vapers of all levels for the past 14 years. We believe that the current range of vaping products successfully caters to a diverse community with varying preferences and needs.
It is our firm stance that introducing further restrictions on flavours and products is not the solution to the issue of youth vaping. Existing legislation is already in place to prevent sales to minors. What we need now is diligent monitoring of retailers to ensure they adhere to these rules, and strict enforcement with fines for non-compliance.
We urge each and every one of you to participate in this consultation and make your voices heard! Your input is crucial in safeguarding your right to choose the vaping products that suit you best. Let us stand together and ensure that adult vapers are not penalised for the shortcomings in enforcing existing regulations.
Please take a moment to respond to the consultation page at https://consultations.dhsc.gov.uk/65201ed1f3410a69990d3081 You can do so anonymously if you wish and it only takes about 15 minutes to complete.
We'll be sharing this information on our Socials and we encourage you share too and help to spread the word that vapers' voices are needed to protect the products we use to stay smoke-free.
Thank you for your support in this matter.
Lisa & Keith
PayPal bans vape sales
Over the last few weeks, PayPal has been busy shutting down PayPal accounts associated with Vape companies. Today it was our turn, after 11 years using their services, they shut down our account without warning!

Fortunately, we'd already been preparing an alternative payment option which we were able to launch within hours of PayPal closing our account so out with the old, in with the new WorldPay option.
Unfortunately our card payment option cannot be used by individuals in the United States so our valued American customers will need to pay for their order via a bank transfer. We highly recommend TransferWise for secure international bank transfers. Their service is quicker, with lower fees and much better exchange rates than most banks and card providers. TransferWise offers the option to fund your transfer by various methods such as credit or debit cards, Apple Pay if available on your device, or you can send a direct transfer to their local bank account. Many other international customers may also wish to use the Transferwise service, to take advantage of the better exchange rate and lower fees than offered by most card providers so you may want to check if they are available in your country.
Don't hesitate to Contact Us if you have any issues using the new card payment option, or setting up a bank transfer payment.
USA's knee jerk reaction to ban flavoured e-liquids
We're sure you haven't been able to avoid the recent media storm with sensational headlines such as "vaping kills" and friends and family might be bugging you about your "dangerous habit".
We attempt here to explain what's actually going on and to put your mind at rest.
Why are they reporting vaping is dangerous?
The term vaping has been associated with nicotine delivery for years, but there are also other substances that can be delivered by a vapouriser such as THC from marijuana. Several states in the US have legalised the sale of marijuana and this has led to a huge increase in the number of THC products. In recent months there have been hundreds of people hospitalised with severe breathing problems and sadly a few have died. Most of these patients have admitted use of THC 'vape' cartridges purchased from the street and we're betting the remaining cases are simply not willing to say they were using black market THC cartridges.
Why is 'vaping' THC a problem?
People have been vaping marijuana in its raw form for many years without issue by heating it in a device specifically designed for this purpose. However, THC containing e-liquids for use in standard vape equipment have recently begun to appear on the US market. Some of these new THC 'vaping' liquids contain diluents and/or additives which are oil-based such as vitamin E (used as a thickening agent). These oils coat the lungs, directly leading to a condition called lipoid pneumonia which causes severe breathing problems and in some cases, death.
So what does this have to do with e-cigarettes?
Absolutely nothing! Let me repeat that - the illnesses and deaths currently being reported are not linked to standard e-cigarettes and are nothing to do with the products we all enjoy. E-cigarettes were designed to deliver flavoured nicotine as an alternative to traditional combustible cigarettes, and typically contain just nicotine, flavourings and the diluents (carriers) Propylene Glycol and Vegetable Glycerine. Any other ingredients or additives are not part of the formula for standard e-cigarette liquids. E-cigarettes are regulated in the UK by the MHRA through the Tobacco and Related Products Regulations (TRPR) and liquids containing nicotine require comprehensive testing before being placed on the market. Unnecessary and potentially dangerous additives are explicitly banned by this legislation. Rest assured we do all we can to provide only the highest quality liquids that have been properly lab tested and found to be free of potentially harmful substances, even those which aren't directly covered by the TRPR such as zero nicotine shortfills.
Why the confusion?
Unfortunately the FDA has been slow to regulate the e-cigarette industry in the US and this has left the market wide open for backyard producers to place all sorts of dodgy products on the market such as these THC vapes. Media outlets haven't understood the distinction, and are attributing these health issues to vaping in general, rather than to a small subset of unprofessionally produced, contaminated products.
So why are they banning flavours?
To cut a long story short, across parts of the US, there have been some issues with the sale and supply of both e-cigarettes and these THC vapes to minors. The problem has been massively exaggerated, and Clive Bates has written a very comprehensive explanation on the topic of the so-called "Youth vaping epidemic". The authorities there have thrown logic out of the window and concluded that teens are attracted to vaping purely because of the flavours, and that banning all flavours except tobacco will stop youth vaping. They seem to be unable (or unwilling) to comprehend that adults like nice flavours, and no longer want that ashtray taste or smell and naturally tend to gravitate towards sweet, fruity and dessert type flavours. We aren't aware of any companies specifically targeting youth vapers with particular flavours, and many vapers cite the availability of a wide range of palatable flavours as the very reason they were able to move away from traditional cigarettes.
Ulterior Motive?
At this point, it might be interesting to explore alternative reasons why they might want to implement this nonsensical flavour ban and make these life-saving products less attractive and less pleasurable for adult vapers in the US. Who stands to lose if vaping continues to grow in popularity as it has done in the last 10 years? The government, losing all those tax dollars from cigarette sales? Big tobacco companies who are not yet heavily invested in the e-cig industry and are seeing their profits decrease as vaping grows in popularity? What about the medical and pharmaceutical industries? Millions of people are no longer smoking and are less likely to suffer all the long term health effects or need life-saving treatments and long term medication regimens.
Vaping hits the medical and pharma industries where it hurts, and it's important to note that these industries are much bigger, more profitable, and enjoy much more lobbying power in the US than they do here in Europe.
Is it just a happy accident for these parties that vaping in general has been confused with the effects of vaping black-market products, or has the information been disseminated deliberately in such a way as to cause this confusion?
Repercussions
We are truly saddened by the action the US have taken. The direct result will be that vapers in the US will be unable to legally obtain the supplies they need to keep them off cigarettes and the number of those making the switch will be reduced.
Vapers across the world are experiencing pressure from friends and relatives who are understandably confused because the news stories appear to be about vaping in general and no-one in the mass media is properly communicating the actual story. Very worryingly, we've heard that some vapers have already gone back to smoking as a direct result of the scary media reports over the last few days.
There is a risk that we may see these same flavour bans spreading across to us here in Europe despite the fact that the Royal College of Physicians have declared vaping to be at least 95% safer than smoking. Politicians don't understand that these dangers are not linked to vaping as a whole and are facing increasing pressure to implement further unnecessary restrictions on vaping products.
We are already stuck with some nonsensical regulations which have harmed the vape industry in the UK (tank size limit, bottle size limit and nicotine strength limit) and it would be enormously harmful to further limit consumer choice if we copied the US in their trend of legislation based on fake news.
Further reading:
The emotional and irrational hysteria in the US about the “vaping-related” lung disease... - Dr. Farsalinos:
http://www.ecigarette-research.org/research/index.php/whats-new/2019/274-us-lung
The great American vaping panic - Christopher Snowdon:
https://www.spiked-online.com/2019/09/11/the-great-american-vaping-panic/
The great American youth vaping epidemic. Really? - Clive Bates:
https://www.clivebates.com/the-great-american-youth-vaping-epidemic-really/
The FDA have come out with the truth yet we aren't hearing this in the media:
https://www.fda.gov/consumers/consumer-updates/vaping-illnesses-consumers-can-help-protect-themselves-avoiding-tetrahydrocannabinol-thc-containing
Clearing up some common myths about e-cigarettes:https://publichealthmatters.blog.gov.uk.....
Nicotine salts - a quick explanation
Nicotine occurs naturally in tobacco leaves in the form of a salt. In this natural form, the active nicotine is not easily "freed up" so it can't be delivered to our bodies very effectively.
In the 1960s, cigarette companies developed a process which added ammonia to the natural nicotine salts from tobacco leaves which made the nicotine more bio-available and provided more of a nicotine hit without increasing the dose. The process they pioneered gave us freebase nicotine, which is the type of nicotine most commonly used in e-liquids. It is also used in nicotine replacement therapies such as patches, gums, lozenges and sprays.
Due to its potency, freebase nicotine provides a harsh throat hit at higher strengths. When it’s used in a high-wattage setups, it tends to be very uncomfortable on the throat, which is why vapers using sub-ohm setups tend to use much lower nicotine strengths. This low concentration prevents the e-liquid feeling too harsh on the throat when inhaled but these low strengths mean vapers have to vape for much longer durations to be satisfied.
Recently, a new chemical process was developed by the vapour industry to enable natural nicotine salt to be more bio-available and to more closely mimic the sensation of smoking a real cigarette. It utilises benzoic acid which helps nicotine salt vapourise at lower temperatures and makes it more absorbable like freebase nicotine. It also delivers the nicotine with a much smoother throat hit.
This new development allows users to obtain a nice potent hit of nicotine in a shorter time using a more portable low wattage device without the harsh throat hit.
Today's pod systems are ideal for salt based nicotine as are most of the standard higher resistance (1.5ohm plus) MTL tanks on the market. Keep in mind though, you should avoid using high powered devices because using sub-ohm devices to vape salt nicotine at high strengths will give you excessive amounts of nicotine which could cause unpleasant side-effects.
Review & Win!
Review any product you've purchased at Creme de Vape to be in with a chance of winning £25 to spend on the site.
How to submit a product review
If you have an account, you can find quick links to review products you've bought within your order history. Alternatively, you can visit a product page and follow the link under the product name to write your review.
Your product review will consist of a rating from 1 to 5 stars (5 being the highest rating), a title, and a description of your experiences with the product. Useful elements to mention in your review could be information about quality, usability, usage tips, pros and cons of the product etc.
Each published review will count as one entry in the prize draw so the more genuine reviews you write, the more chances you have to win.
Review guidelines
Your review must be your own genuine thoughts about the product you have purchased at Creme de Vape.
Copying opinions that have already been published elsewhere is forbidden.
Multiple reviews of the same product will not qualify as multiple entries into the monthly prize draw.
Multiple reviews of different products using the same or very similar text will not qualify for entry into the monthly prize draw.
Reviews are not the place for questions, conversations, mentioning other products or websites etc. Please keep it clean and to the point and make sure it's about the product itself.
Emojis are not supported at this time.
We reserve the right to remove reviews from the site if they do not meet these guidelines.
Prize draw terms
On 15th-17th day of every other month, each eligible review since the previous draw will be assigned a unique number and the winning entry selected by a random number generator.
The prize will be in the form of a voucher to be used at Creme de Vape.
The voucher will be valid for 6 months from the date of issue.
The voucher must be spent in a single transaction and is not valid on postage and packaging costs.
If you would like to place an order that is worth more than your voucher amount, you can pay the difference using PayPal or a credit / debit card.
If the voucher is worth more than your order, the difference will not be refunded; therefore if you do not use the entire voucher amount, the difference will be lost.
The voucher amount advertised includes VAT, so if you are outside the EU, the voucher value you will receive will be £20.83 ex-VAT.
The voucher has no cash value and cannot be exchanged for cash.
The writer of the randomly selected review will be sent a private email and announced publicly (first name and town/country) on this site and on social media.
Why is my tank leaking?
A leaking tank is something we've all experienced, but it's usually pretty easy to fix if you can identify the reason for the leakage.
Leakage happens when your atomiser is flooded and the e-liquid will usually find its way out of the airflow system. If you've got a tank which is flooding, gurgling or leaking, you'll have a poor vaping experience, and you'll want to try to fix it because it could harm your device or cause it to fail if it reaches the electronics.
The first thing to understand is that tanks and heads designed for Direct Lung (DL) vaping are generally designed for a thicker, higher VG liquid and usually feature larger wicking holes to wick liquid faster to the coil to keep up with higher coil temperatures/wattages. If you're using too thin a liquid in a DL tank, you are likely to experience leaking. Mouth-to-lung (MTL) tanks usually have smaller wick holes, are used at much lower wattages and tend to wick juice slower to avoid flooding. Typically most MTL tanks will not cope with a very high VG liquid because of this. So if you're using the wrong liquid for the type of tank or head, you're likely to experience either leakage, or the opposite - poor wicking and dry coils prone to burning.
Other than trying to use the wrong liquid or tank for your vaping style, common reasons for flooding include:
- Incorrect tank assembly or damaged/missing parts
It sounds obvious, but it's important to ensure your tank and head are all put together correctly, because improper assembly commonly causes leakage. If any of the parts are cross-threaded, improperly assembled, damaged or cracked, or if any of the seals are damaged or missing, it will cause leakage.
- Liquid or condensation in the chimney
On some tanks, it's easy to accidentally get juice in the central airflow tube which leads from the mouthpiece down to the coil when re-filling. Sometimes cooled vapour condenses here and some tanks need the inside of the chimney cleaned regularly. If you find you are experiencing a lot of condensation build-up, try altering your vaping style a little, by taking your finger off the fire button a bit before you've finished your inhalation. This should ensure the entire vapour production from that activation has been inhaled and no vapour residue has been left in the chimney to condense back into liquid.
- Your e-liquid is a little too thin
If your liquid has a higher percentage of PG, you may want to move to one with a bit more VG, or even add a little VG yourself. This should slow down the wicking and hopefully be a little more effective at preventing flooding. This is especally important if using a tank designed for DL vaping, as the wicking holes in the coil are usually much larger so thinner liquids will leak through quicker.
- The coil needs changing
Most vapers find they get 5-10 days of good use from a coil depending on their vaping frequency and style. Your mileage may vary, however, you will want to change your coil whenever you notice a change in taste, vapour, performance or wicking, and a flooded or an overly dry coil can both be signs that the coil is on its way out.
- Filling technique
Some top-fill tanks are more prone to leakage during or after filling. Every time you open a top-fill tank to fill it, the pressure is changed inside the tank, and tanks which have a top cap that is completely removed to fill can be prone to trapping air and pushing more liquid into the coil when you screw the cap back on after filling. Some manufacturers have cleverly designed caps which work to prevent this and others have created innovative liquid control solutions which allow you to completely close off the liquid feeding to eliminate flooding when the cap is opened. If you have liquid control, close the liquid feeding off - if you don't, then try closing the airflow off before you open the tank to fill it - this helps in many cases, but this isn't a foolproof fix. Get into the habit of leaving a little pocket of air at the top when filling rather than trying to fit the maximum amount of liquid in the tank.
- The airflow setting might be set too tight
If your airflow setting is too restricted, this can create a stronger suction effect, which causes the tank to feed liquid to the coil faster than it can be vaporised. If your tank has adjustable airflow, try opening it up to an airier setting to see if the flooding reduces. You may be "pulling" a bit too strongly - in some tanks, the airflow setting and the strength of the suction can both cause too much liquid to be brought to the coil, thus promoting flooding. Try a softer draw or an airier airflow setting.
- Inappropriate power setting
You might not be using a high enough power setting to vaporise all the liquid coming to the coil. This is particularly pertinent if you are using a tank designed for DL vaping. Coils below 1 ohm are mostly used for DL vaping, at higher power (usually above 25W), with thicker juice and with unrestricted airflow. If you're using a power setting lower than is recommended for the coil you're using, it may be feeding more liquid to the coil than can effectively be vaporised. Raise your power setting, or switch to a higher resistance coil.
- Improper wicking (rebuildables only)
If you're using a rebuildable atomiser, you'll need to ensure you're following the manufacturer's recommendations for wicking. Sometimes working out the ideal wick size and placement takes a bit of trial and error to get the best results for your particular atomiser and vaping style.
- Tank has been left for too long between uses
Most tanks will continue to wick the juice to the coil even when the tank is sitting idle. If you don't use your tank frequently, you'll usually find it floods more easily. If you're going to leave your tank unused for a couple of days or longer, it's best to empty it out and use a new coil next time. Some tanks (with a bottom coil and airflow) can be stored upside down for a few days with e-liquid in them to prevent wicking and flooding, but it's not recommended for more than a few days as you don't actually want your coil to completely dry out between uses.
- Tank has been left on its side
Some tanks are much more prone to leakage if they are left on their side between uses, or overnight. Try to keep your tank upright during vaping, and when its not in use.
What to do once your coil has flooded or leaked
Different methods work for different tanks - try one or more of the following to recover a flooded head.
- Take your tank off the device, and sharply blow down the mouthpiece to push the excess liquid out through the airholes into a tissue.
- Roll part of a tissue up and insert it into the mouthpiece and invert the tank to try to clear the flood out of the top. Best to also clean the inside of the chimney after this method with another piece of rolled-up tissue.
- If the coil head sits inside a base, take the head out and use a tissue or cotton bud to clean out the base. Remove any visible excess liquid on the outside of the head and also clean the inside of the chimney with a rolled-up tissue.
Some heads are just more flood prone than others, particularly if you have quite a heavy “draw” and some may continue to flood even after blowing out, especially if the tank or coil isn't suited to your vaping style or your power settings are outside those recommended by the manufacturer.
How long does an atomiser coil last?
How long should an atomiser coil last? How long is a piece of string?
Pinning down the eternal coil longevity question is pretty much impossible, because of the different variables involved.
Certainly as a beginning step, it's more worthwhile to think of coil longevity in terms of the amount of e-liquid that's been vapourised, rather than a period of time, because the amount of juice that's been through a coil is more of a valid measure of coil life, although it's not the whole story.
Vaping style
Whether your tank and your vaping style is mouth-to-lung (MTL) or Direct Lung (DL) will usually affect how long your coils last.
MTL vapers use higher resistance coils on much lower power settings, for a cooler vape which uses less liquid, therefore, MTL coils typically last longer than DL coils, but not always.
DL vapers use much lower resistance coils on much higher power settings. This higher performance and much greater vapour production is very desirable but it does mean that more juice is used and typically leads to a shorter coil life.
Other aspects of vaping style include how long you activate your device for each time you take a puff, and how strongly you "pull" on the mouthpiece because this alters the airflow and the airflow can affect the longevity of the coil.
Power
This is related to vaping style. If you are a DL vaper, you're running at a higher wattage, and the coil burns hotter, placing extra demand on the wick which can burn and produce a foul taste if the juice isn't wicking through quickly enough to meet your vaping intensity. Of course, higher power often means more juice, and as already mentioned, the longevity of a coil is mostly related to how much juice is vaprourised through it.
Juice type and strength
We've found that some juices have a tendency to "clog up" the coil more quickly than others. This is a case of experimenting to find what works for you. Sometimes it's worth putting up with a slightly shorter coil life to enjoy that fabulous flavour you love. Some say that higher VG juices clog up the coil more, but this could be more to do with the fact that higher VG juices are typically used by DL vapers who go through more juice than MTL vapers.
We also commonly see liquid consumption increase when people lower the strength of the e-liquid they use. This can then have an impact on coil life as more juice is consumed.
Device
Modern devices tend to perform better and vapourise liquid more effectively. The net result of better performance and better vapour production is that you go through more e-liquid - that's the nature of the beast. If you want a better vaping experience, you will generally use more juice and coils than you would with an older or less powerful device which struggles to keep you satisfied.
You may have been used to a longer coil life on an older device which was less powerful and less effective or perhaps your old device had dirty or oxidised contacts or a battery which has been charged and discharged so many times and it's just not as effective any more.
Type of tank/coil
It's important not to forget that every tank and every coil is unique and your usual coil longevity will vary from tank to tank even when all other factors remain the same.
Improved taste perception
As time goes on, vapers do tend to develop a greater perception of taste changes in the coil which leads to the necessity to change the coil more frequently than you might have done originally. When you're just moving away from cigarettes, your taste perception can remain dulled for some months and it's only when it really starts to return over time that you notice the coil starting to taste bad sooner than before. Most people experience a shorter coil life the longer they've been vaping (stock heads as well as DIY coils). We sometimes receive reports that the coils seem to have changed and don't last as long as they used to, but it's pretty rare for manufacturers to change anything about the materials or manufacturing of existing coils, and these variances are almost always down to one of the factors detailed in this post.
Increased satisfaction with vaping
We've found that increased satisfaction with vaping, whether due to a new kit or a new flavour, sometimes leads to an increase in vaping and in juice consumption. People often struggle along with old, poorly performing hardware far longer than they realise, and then when they upgrade, they suddenly notice they have to replace their coils more frequently. This can be due to the better performance of a newer device, or an increase in vaping because it's just a much better experience than it was with the old kit.
Other considerations
Some people believe that different climate conditions and temperatures also play a part. Some liquids don't feed very well in some tanks (particularly thicker liquids in MTL tanks) during the colder months, so this will lead to wicking issues, and a poor taste as the wick struggles to keep saturated.
How to extend the life of your coils
There are things you can do which can help make that new coil head last a little bit longer. The first step is to prime your coil. The idea behind priming is to try to saturate the wick within the coil head before first use, to reduce the chances of burning especially if you're impatient and want to use it right away without waiting for 10-15 minutes for it to wick naturally.
- Method 1
There are usually juice holes on the side of the head which is where the juice from the tank feeds into the wick. You can carefully place a few drops of juice onto each hole (let each drop sink in before dripping again) and a few drops into the top of the head (avoid the airhole in the middle of the head). - Method 2
You can try taking a few drags from the tank without firing your device. This can be done 4 or 5 times to try to encourage e-liquid to feed through to the coil. - Method 3
Similar to method 2, but generally works better. If your tank is one that has adjustible airflow, close the airflow down completely and do 2 or 3 strong pulls without activating your device. This method saturates the wick more effectively than method 2, however it is quite easy to flood the coil if you overdo it. With experience, you'll learn to sense when the wick is saturated and to stop before it's flooded.
Once you have the coil sufficiently saturated, it's a good idea to break it in gently by using a lower wattage for the first few hits. Then gradually work up to your normal wattage once the new coil has bedded in.
Many people swear by these processes because they enable you to use new coils right away without risking burning them due to a dry wick.
My own experience
I use a mouth-to-lung tank at 13 watts and I usually get somewhere around 120-180ml through a head before I have to change it because it's tasting bad or feeling clogged. This is pretty much universal for me across most MTL tanks.
I use method 3 to prime my coil, so I can start using it straight away without risking a burnt coil. If I am vaping too intensively and the tank is struggling to keep up, I again use priming method 3, just once, to bring more juice to the coil to cope with my intensive vaping.
We'd love to know what setup you're using and how long a coil lasts you.
Lisa
Welcome to our new site!
We are thrilled to announce the launch of our new website!
We have been working behind the scenes on a new look and feel for our website and we are very excited to finally launch it. Don’t worry, all your details and order history remain easily accessible and you won’t even have to change your password.
Desktop and tablet users will notice that the navigation has been moved to the top of the website. Some products have moved to new categories to make them easier to find (we hope). If you're stuck, the product search box is more prominent and works fabulously.
The simple checkout so many of our customers appreciate remains as easy to use as always.
Our original site was designed long before mobile devices were in such heavy use for internet browsing and online shopping and we have worked really hard to improve the experience for all. We hope you will find the new layout much more usable on your mobile devices and it provides a cleaner look on tablets and desktops too.
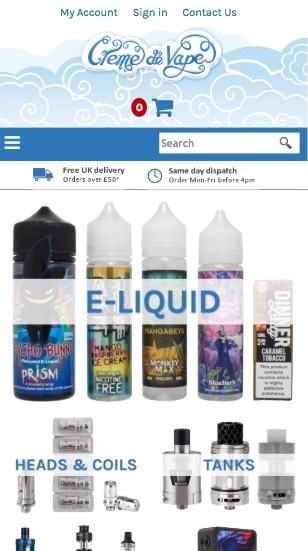
Below is the new mobile view
The E-liquid category has been completely overhauled making it much easier to find your preferred type and strength, and our new e-liquid flavour selector (available on the home page and in the e-liquid category) makes it easier than ever to find your perfect vape.
Mobile product filtering
Product filtering is now available on mobile devices. Simply touch the "Filter Products" bar to open or close the filter options for the category you are in.
We have done a lot of internal testing, but should you come across anything you think looks weird or is not working for you, please let us know so we can fix it.
We hope to serve you very soon.
Keith's take on temperature control
I’ve been enjoying temperature control for over 4 years now and while it did take me a little time to get my head around it all, once I understood what’s going on with the technology and use that temperature control setting as a scale, not as an actual reference to a real temperature.
In 2014 Evolv was the first to market with their temperature control feature in the DNA40. As with all new tech I was eager to give it a try. It worked and did what it claimed to do albeit buggy and in need of some refinement, but I feel there was a bit of a flaw with the terminology used in setting your desired heat level. Sure, one can work out what the approx. temperature of a given wire should be based on the manufacturer’s specification for the resistance changes at different temperatures. This is called the TCR or temperature's coefficient of resistance. In simple terms this means that as a resistance target (the temperature setting) is reached on the TCR scale we can calculate the approx. temp of that wire based on the change in resistance from the room temperature's resistance.
But here’s where I think the terminology (but not the functionality) is flawed. We are not using an isolated straight piece of wire alone, we are using a coiled piece of wire attached by two screws or clamps with some sort of wicking material running down the middle with both liquid and air passing across it. Thus an actual temperature value is never going to be entirely accurate nor consistent - there are simply too many other variables at play when it comes to that "temperature" value.
Think about the difference between an oven and a hob (stove top). An oven usually has dials so you can set a desired temperature, this is easy for the oven's electronics to control because inside the closed space there is a thermostat (a device that measures the temperature) and this sensor is used to help regulate the power applied to the heating elements (coils) inside the oven to maintain that set temperature. Now take a look at the hob controls, these are normally dials or buttons that have a scale (ours goes from 1 to 10). There is a good reason why the hob has no temperature settings on those dials. There is usually no means for the electronics to measure the actual temperature of that particular heating element on the hob. This is also true with our vaping devices, there is no sensor to measure the actual temperature inside the atomiser's chamber. Therefore, I think it may have been better to use a scale from 1 to 10 or 1 to 20 for this setting on a TC device, but this is not the direction the market took and we have to understand the quirks of this technology and understand what’s actually going on.

Factors that play a role in the TC experience:
TCR value, every wire material type has a recommended value. This value is what the manufacturer of that wire states the resistance change is at a given temperature. In our vape devices we are using a max temperature coefficient of around 600F. The TCR value is how much change in resistance it takes to go from room temperature to a max of 600F. When you set your "temperature" on the device you are not actually setting a temperature, you are setting a resistance target on that TCR scale. Wire materials are not 100% consistent across all batches, all manufacturers etc etc... This is an approximation only and should only be used as a reference value to start with and most all devices on the market have the ability to set a custom TCR value, use this feature if you need to, do not rely on a wire type setup in the device from the factory as the default only. Personally I find that Ni200 having the highest change in TCR works best for temp control as there is a greater change in resistance as the wire heats up. Low TCR value wires such as stainless steel are not as stable and can fluctuate as the device attempts to work out the setting on very small changes which leads to a jumpy temperature that you can actually feel as you vape on some devices. The TCR value is based on the wire ITSELF, not all the other external factors such as liquid hitting it or air passing over it.
Wicking, this is a big one. As the temperature control device attempts to read the resistance change there is (or should be) a constant flow of liquid hitting the wire which in turn cools it down again and the device is able to hold a desired setting while you vape. If your wick/material or atomiser is not keeping up with the heating element the resistance climbs quickly and the power is reduced to maintain your target resistance (temp setting) and this leads to a weak vape -by design- keeping you from over heating the wick and getting a burnt taste from your juice. Too much liquid flow and things can go the other direction resulting in flooding and spitting as you actually get some boiling of the liquid. There is fine balance here between not enough liquid to the coil and too much. You just have to gain experience with this area and know your atomiser's ability to keep up with the wicking required in TC vaping. Don't be scared to experiment with different wick materials and amounts of wick you install in the coil. Some materials are better at keeping up than others, and what works for your friend may not work for you and your application. Every juice also has a different viscosity at different temperatures so bear in mind on warm days you may experience different wicking than you would on a cold day.

Air flow, another big factor. The amount of air passing across the coil cools it down significantly and lowers it's resistance. But air flow also has another role to play with an RTA. The amount of air pressure you apply by puffing on the device compared to the air hole size actually pulls liquid into the wicks at different rates. Every person puffs on their device with a slightly different pressure every time they vape and this can actually change though the day too. It's just not possible for a human to pull the exact same pressure on the mouthpiece every puff, all day long. As the market moves back to the mouth-to-lung style vape we are finding that the puff pressure has been decreasing a bit as the air hole sizes decrease (this was the norm 5 years ago). When you are vaping on an atomiser with large air holes and you are taking direct lung hits there is a lot of air passing the coil keeping it cooler and also pulling more liquid to the coil which can result in a warmer vape. With an MTL setup you may experience a weaker vape with TC as less air is passing the coil and less liquid is entering the chamber, you will need to experiment with the settings to get your desired vape, remember what I said before and think about temperature control as a sliding scale rather than a set of actual temperature values and bear this in mind when you are setting up your temp control device.
Connectivity. Every mod has a 510 connection on the top where you screw your atomiser down onto. This is a weak point for every temp control system. The threads are where the negative side of the circuit passes current. If your threads are dirty or wet with liquid (on either the mod OR the atomiser side) this can lead to an inaccurate temperature control experience. Make sure your threads and the 510 centre post on both the mod and atomiser are very clean and dry at all times to avoid issues of varying resistance (often called "Jumping"). If you knock or drop your device it's always a good idea to double check your settings and re-calibrate your coil to be sure everything is normal.

I hope this helps you understand a little better what may be happening inside your temp control device. Don't be too quick to jump to conclusions that your mod or atomiser may be faulty if your TC vape experience is not what you think it should be as there could very well be a small detail that's causing an issue for you. Don't be afraid to experiment with different wick materials, atomisers, or wire types. You may just find your perfect vape. What works for you may be completely different to what works for me. For temp control vaping, my favourite device is the Dicodes as it offers a stable and accurate experience. The room temperature resistance of the coil is locked by the user and the display on the firmware provides the ability to check at any point in time what the calculated resistance and temperature of the coil is (R/T menu) This gives me the ability to double check the coil at room temp to be sure nothing has changed or if it needs a recalibration, this is not a feature available on any other mod on the market (without hooking up to a PC and using software anyway).
Shortfills have arrived
Well, finally, shortfill "shake and vape" e-liquids have arrived at Crème de Vape. They provide a cost-effective way for vapers to obtain larger bottles of e-liquid and avoid having to deal with all those little bottles.
Why has it taken so long for us to get them in?
Good question - I'll need to provide a bit of background to answer it.
We noticed shortly after the end of the TPD transition period in May 2017, retailers started offering large bottles of flavoured e-liquid without nicotine, with empty space for TPD compliant flavourless nicotine liquid to be added by the consumer because the TPD/TRPR regulations do not currently apply to liquids without nicotine. This has been seen by many as a positive development, however, it was unfortunate that so many producers rushed in to take advantage of this loophole with all manner of completely untested liquids.
We've spoken to many companies over the years who simply don’t care to test their product and seem not to be bothered at all about what their customers are ingesting. I can’t count the number of times that our request for test results has elicited a response along the lines of “no-one else has asked” or “you're the only one with a problem”. These new untested shortfills took us back to how the e-liquid industry used to be pre-TPD when we faced extreme difficulty getting any kind of information about product content from manufacturers, and we had to run our own testing because we refuse to stock potentially unsafe liquids.
The industry had been aware of the problem since Dr Farsalinos' 2014 report where he revealed he'd tested and found potentially harmful ingredients in a lot of e-liquids on the market. He tested for Diacetyl (2,3-Butanedione) and Acetyl Propionyl (2,3-Pentanedione) which are buttery/creamy flavoured chemicals which have been linked to permanent lung damage when inhaled. See his study report here. Consumers and manufacturers were starting to sit up and take notice.
We put our cards on the table some years ago (when we were known as Cloud9Vaping) when we publicly blogged about our long-standing e-liquid testing policy (see our blog post here). We stated we'd removed a range of e-liquid from sale shortly after commencing stocking it, because our testing revealed that contrary to the manufacturer's own assertions, the liquids contained very high levels of these potentially harmful ingredients. Many customers asked us which range we had removed from sale, so eventually we revealed the full story which lead to a huge debacle much of which was publicly played out. The manufacturer then threatened us with legal action for revealing information that they already knew about, but had concealed from retailers and consumers.
Gradually, the industry started to listen and vaping trade and consumer associations also started to recommend avoidance of liquids with these ingredients. Manufacturers started to actually recognise the problem and many began to take steps to deal with it and reformulate their liquids.
When the TPD/TRPR arrived, it mandated emissions testing and full ingredient disclosure, along with toxicology reporting for every single ingredient present. It also completely disallowed certain ingredients including the two I’ve mentioned above, which was a great step forward even though many of the other measures in the TPD have been ill-advised. See the TPD UK ingredient guidance. We thought it would finally rid the market of all poor quality liquids including those made with inappropriate ingredients that shouldn’t be inhaled – however that has not been the case as many producers of zero nicotine liquids have made no effort to perform any testing or comply with the banned ingredient list
The four ranges of shortfills we now stock have all been tested in exactly the same way as TPD compliant e-liquids, and none of them contain any of ingredients banned by the TPD, with the exception of Blackberry Crumble by Dinner Lady which contains a trace amount of naturally occurring (unavoidable) Diacetyl which is below the AFNOR recommended maximum level.
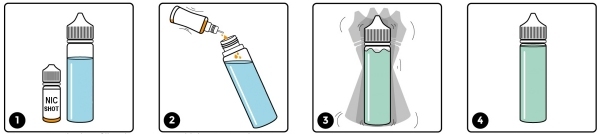
It’s possible that the unregulated shortfill market will be shut down at some point in the future when the regulators take notice and close this loophole. We expect the authorities to mandate that they be tested in exactly the same way as any other e-liquid and we would welcome this change. We only have to hope that they don’t put the 10ml limit on these liquids as well – if they do, it will be entirely because of those who tried to avoid testing and are happy to supply unknown substances for unsuspecting consumers to ingest.
Useful links
Tobacco and Related Products Regulations (TRPR)
TPD UK Ingredient Guidance
TPD Update
We understand there is still some confusion about the EU Tobacco Products directive (TPD) which came into law across the EU on May 2016. The UK implementation is known as the Tobacco and Related Products Regulations 2016 (TRPR). It contains various restrictions and regulatory burden all of which will affect our customers to varying degrees. We’re currently in the transition period, and we can sell existing stock without restriction. This period ends on 19th May 2017, after which all products sold must meet the new requirements of the TRPR. Liquids without nicotine are exempt from these requirements.
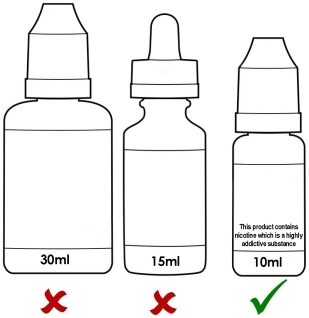
Maximum 10ml bottle size and maximum nicotine strength of 20mg/ml (2%).
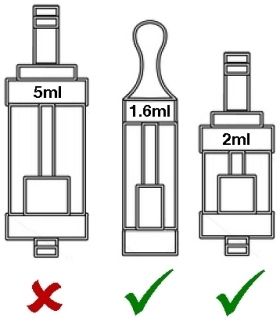
Maximum 2ml capacity for tanks, which must have a leak-free filling mechanism.
In addition to the size and strength restrictions, where a product contains nicotine, or could be used to contain nicotine the TRPR requires:
- A large warning about nicotine on the front and back of the packaging (even where the product is sold empty).
- Ingredients, composition and additional information and warnings to be on the outside of packaging and on enclosed leaflets.
- Products to be comprehensively tested (including their emissions during use) and the data notified to the governing body 6 months before products are placed on sale (or before November 20th 2016 for products already on the market).
- Sales of each notified product reported annually.
Some of the products you rely on may change to a compliant format, but many will not, and will become unavailable for sale to consumers in the UK and EU. The substantial costs involved in the testing and notification will lead to a further reduction in the variety of products on the market as most manufacturers will only be able to afford to test and submit their best sellers. Prices may increase slightly to cover the costs of the testing and notification, as well as the additional packaging.
We have been unable to import any non-compliant products since November 20th 2016, and so non-compliant products are beginning to sell out, and won’t be replenished. As you're browsing the website, you may notice a red box with the text "NOT PURCHASABLE IN THE EU AFTER MAY 19 This is your cue to buy now, whilst you still can, because you will not be able to purchase this product after this date. If you see this on a 30ml e-liquid, we recommend you browse the site to see if there is a 10ml compliant version available.
We advise you to ensure you have stocks of your favourite products on hand well before May and transition to compliant products as soon as they become available. We have been concentrating our efforts and working hard with our manufacturers to prepare compliant items over the last 6 months and although there are huge challenges to overcome, we are hopeful about the future of vaping post-2017.
For further information please see our earlier TPD blog post.
UK Vapefest 2016
UK Vapefest 2016 draws ever closer. It's a chance to catch up with all our friends, and have a lovely day out with fellow vapers.
Make sure you enter the raffle for a chance to win the amazing prize below.
Come see us on Saturday 6th August at:
The Greenhous West Mid Showground
Berwick Road
(Entrance on Gravel Hill Lane)
Shrewsbury SY1 2PF
Please contact us to let us know if you would like us to bring anything in particular with us.
We very much hope to see you there.
Prize contains the following:
Vapefest Special Edition Dicodes No.6, SvoeMesto Kayfun 5, Efest 26650 battery x 2, Efest 26650 battery case, XTAR VC2 charger, UD Coil Mate Tool Kit, Fiber Freaks XL Pack Cotton Blend, Dicodes Resisterm NiFe30 Wire, UD 24ga (0.5mm) SS316L wire, 30ml Njoy Artist Collection 6mg/ml e-liquid x 5, Crème de Vape Microfibre pouches, Crème de Vape Vapebands, Mini screwdriver keyring, Keyring flashlight.
Prizes are not refundable or exchangeable.
PHE Issue new guidance on vaping in public
Public Health England (PHE) have released some very welcome guidance about vaping policy in public and in the workplace.
Their advice is measured and sensible throughout and they repeat that vaping carries a fraction of the risk of cigarettes and has the potential to help drive down smoking rates and improve public health but that policy makers must make clear the distinction between vaping and smoking. PHE is strong in its assertion that the estimated 2.8 million vapers in the UK need to be encouraged to stay smoke-free and that future policies about vaping in public should be focused on fostering an environment in which e-cigarettes can provide a route out of smoking for England's eight million smokers.
There are many positive points within the guidance including a welcome acknowledgement that smokers can achieve their desired nicotine level much quicker than vapers, and this difference should be taken into account particularly in workplaces.
We thought this statement was most notable and urge you to share this with your employer if you are currently being forced to vape alongside smokers in a smoking room or designated smoking area in your workplace:
"it is never acceptable to require vapers to share the same outdoor space with smokers. Where a designated outdoor smoking area has been provided in a public place or workplace, vapers should be allowed to vape elsewhere"
Links to all the documents can be found below.
E-cigarettes in public places and workplaces: a 5-point guide to policy making.
Use of e-cigarettes in public places and workplaces: advice to inform evidence-based policy making.
Report of PHE stakeholder ‘conversation’ on use of e-cigarettes in enclosed public places and workplaces
Lord's last ditch attempt to save vaping
In the House of Lords Lord Callanan has put down a motion which if passed would stop implementation of the TPD in the UK. This is a critical development.
Lord Callanan is a hugely experienced senior politician. In the European Parliament he both advised how to campaign against medicalisation and led the successful campaign to block regulation that would have wiped out e-cigarettes. Nobody has been more effective at defending vaping than him.

Unless Lord Callanan is forced by pressure from Downing Street to withdraw his motion there will be a debate and a vote. Under parliamentary rules if the Lords agree to this motion the statutory instrument implementing the TPD in the UK will fall. Such procedures are called “fatal motions” because the regulations get killed outright and do not go back to the Commons.
(For geeks: the Lords have until 10 June to vote against this delegated legislation. That will be the end of the 40 days of “praying time” during which a successful motion can block the statutory instrument. This is the case even though the law has previously gone into effect as the TPD will on 20 May.)
Politics
The Lords are strongly against the TPD rules on vaping as was shown in last week’s debate in the House of Lords.
Government and Opposition
They know there are severe problems with the TPD’s rules on vaping. Government officials have acknowledged that they could increase smoking and the health minister told the Lords that he hoped enforcement would be more ‘Italian’ than traditional British.
The shadow minister said that he could not understand why e-cigarettes were included in the TPD and described opposition to vaping as “bonkers”. Two years ago his boss, Jeremy Corbyn, signed a parliamentary motion against the TPD rules on vaping.
Momentum
As politicians see increasing vaper pressure they are already trying to be creative in finding a solution. Technically the EU could start “infraction proceedings” against the UK for not fully implementing Article 20, but this would prove bad timing. This is not the time encourage anti-EU sentiment!
Why we will succeed
There are more of us.
In 2013, despite huge pressure from the EU commission, vaper power helped swing the day. At that time there were 1.3 million vapers in the UK. Now there are 2.8 million of us.
Political concern
Westminster and Brussels have reasons to be helpful. With top doctors such as the Royal College of Physicians coming out strongly in support of e-cigarettes, politicians are finally seeing the evidence that some of the rules are no longer fit for purpose.
As we saw last week, a majority of vapers want to leave the EU. Politicians (at least those who are in favour of the UK remaining in the EU) will not want to see vapers decide their referendum votes on this single issue.
What you can do
If ever there was a day for vaper power this is it! Last time round, it was the letters and social media support that won so many MEPs to our cause.
So let us respectfully urge our leaders to use the opportunity provided by Lord Callanan’s motion to rethink the specific problems we have with TPD.
Tweet: send your support for the #LordsVapeVote to @Number10gov @jeremycorbyn @MartinCallanan. (Update: #LordsVapeVote is now trending on twitter!)
Petition: Here is one ready to go, and in just a few hours it has already attracted 1000’s of signatures. Add yours now: Petition to Parliament
When vapers work together they are an unstoppable force. If we do not take today’s opportunity presented by Lord Callanan’s initiative then we could have to wait until around 2026 for the next Tobacco Products Directive to be implemented for even a chance of improvement.
So wait ten years – or tweet today like you have never tweeted before.
Thank you to Ian Gregory of Abzed for much of the content of this post.
Call to Action - Please write to your local MP
Last week we informed you about the Royal College of Physicians (RCP) report on electronic cigarette research, this week we are urging you to please write to your local MP asking them to support an early day motion calling for debate before the UK Parliment about the upcoming Tobacco Products Directive implementation on 20th of May this year.

Parliament UK
Early day motion 1441
E-CIGARETTES AND THE TOBACCO PRODUCTS DIRECTIVE
Session: 2015-16
Date tabled: 04.05.2016
Primary sponsor: Anne Main
That this House agrees with the Royal College of Physicians that it is crucial that e-cigarettes are priced as advantageously as possible in relation to tobacco.
Believes that the EU Tobacco Products Directive would significantly inhibit the development and use of harm-reduction products by smokers and cost lives.
Further agrees with Public Health England that e-cigarettes are around 95 per cent less harmful than smoking, and that nearly half the population does not realise that e-cigarettes are much less harmful than smoking.
Further believes that restricting advertising will have the perverse effect of reducing the rate at which cigarette use is declining.
Notes that the total cost of smoking to society, including healthcare, social care, lost productivity, litter and fires, was conservatively estimated by Action on Smoking and Health to be around £14 billion per year.
And calls on the Government to exclude e-cigarettes and other harm-reduction products from the Tobacco Products Directive.
You can write to your local MP using this website: https://www.writetothem.com/ Please be polite and explain why you feel the TPD will impact your choices for vapour products, and how e-cigs have changed your life. You need to be sure to urge your MP to support the "Early Day Motion 1441"
If you have not read the Royal College of Physicians' report and shared it with your friends and family members, please do so. The report highlights what we as vapers have known all along, here are just a few of the key points of the full report.
- Smoking is the biggest avoidable cause of death and disability, and social inequality in health, in the UK.
- People smoke because they are addicted to nicotine, but are harmed by other constituents of tobacco smoke.
- E-cigarettes are proving much more popular than NRT as a substitute and competitor for tobacco cigarettes.
- There are concerns that e-cigarettes will “renormalise” smoking, but to date, there is no evidence of this occurring to any significant degree in the UK.
- In the interests of public health it is important to promote the use of e-cigarettes, NRT and other non-tobacco nicotine products as widely as possible.
- There is a need for regulation to reduce adverse effects of e-cig use but the regulation should not be allowed to significantly inhibit the development and use of harm-reduction products.
It is excellent news that this comprehensive report fully recognises the importance of e-cigs as harm-reduction, however it may have come too late to prevent the damage that will be caused by the Tobacco Products directive coming into force next month, which will significantly inhibit the development and availability of these products. Be sure you have also read our blog post about the TPD (Tobacco Products Directive).
Breaking news flash - RCP report
Breaking News Flash!
We urge everyone to read the Royal College of Physicians' report and share it with your friends and family members.
The report highlights what we as vapers have known all along, here are just a few of the key points of the full report.
- Smoking is the biggest avoidable cause of death and disability, and social inequality in health, in the UK.
- People smoke because they are addicted to nicotine, but are harmed by other constituents of tobacco smoke.
- E-cigarettes are proving much more popular than NRT as a substitute and competitor for tobacco cigarettes.
- There are concerns that e-cigarettes will “renormalise” smoking, but to date, there is no evidence of this occurring to any significant degree in the UK.
- In the interests of public health it is important to promote the use of e-cigarettes, NRT and other non-tobacco nicotine products as widely as possible.
- There is a need for regulation to reduce adverse effects of e-cig use but the regulation should not be allowed to significantly inhibit the development and use of harm-reduction products.
It is excellent news that this comprehensive report fully recognises the importance of e-cigs as harm-reduction, however it may have come too late to prevent the damage that will be caused by the Tobacco Products directive coming into force next month, which will significantly inhibit the development and availability of these products.
Be sure you have also read our blog post about the TPD (Tobacco Products Directive).
The future of vaping changes in May 2016
You may have heard rumours about upcoming regulations which will affect the availability of many of the vaping products you currently enjoy.
We’ve been hesitant to communicate much of this information directly to our customers thus far, because the situation has not been clear enough to be able to properly inform you about exactly what will happen. Information directly from UK regulators has slowly started to come through but there’s still grey areas and ambiguity in the information released so far, so it’s still quite difficult for companies to plan properly or advise their customers on what the future holds.
Having said that, we know a large proportion of our customers still don’t know anything about these looming changes and we felt it was time to give you a run-down of what we know so far as well as comment on what we don’t yet know. Apologies that this is quite a long post, but it’s a complex topic, and requires as much explanation as we can provide to ensure you are as well-informed as you possibly can be.
On May 20th 2016 The EU Directive 2014/40/EU1 also known as The Tobacco Products Directive (TPD) comes into force. This Directive changes how tobacco and related products are manufactured, marketed and sold within the EU and contains some very restrictive measures relating to vaping products2.
Many feel (us included) that the restrictions relating to electronic cigarettes didn’t belong within a directive relating to tobacco products at all, and that regulatory provisions for these innovative, and life-changing products belong in separate, more carefully considered legislation, which could have ensured vapers continued to have access to the products they need and the right to choose a much safer alternative to smoking. However, the directive as it is was pushed through to further regulate cigarettes, tobacco products, and vaping products, all in one go.
As a result of this legislation, many of the products we know and love will disappear and the products currently available on the market will become very limited. The rate of innovation will also slow due to the financial burdens and delay involved in bringing new products to market, compounded by the 6 month delay that’s been imposed for newly notified products. The regulations do however allow a sell-through period3 for the sale of old stock until May 2017.
Unfortunately, there’s still a LOT up in the air due to the regulators’ failure to unambiguously define what’s included and what isn’t as well as the fact they have repeatedly contradicted themselves on various documents we’ve seen. This uncertainty means that much misinformation is out there, and manufacturers and retailers are not yet able to state exactly what the position will be, even though the restrictions will take effect in just a few months’ time.
We’ve chosen to focus here on those elements of the legislation that will be most restrictive for existing vapers, however there are various other provisions that will dramatically affect vaping. One that is of great concern to those of us who are keen to spread the message of vaping to as many smokers as possible is the ban on all forms of advertising and promotional activity, which may include online discussion forums and specialist vaping broadcast channels as well as traditional print and TV/radio media. Please rest assured that we’re doing our absolute best to navigate our way through the legislation, and working with our manufacturers to ensure they are also up to speed, and capable of producing compliant products. However, these changes ARE coming, and all current and future vapers will be radically affected.
Read on for detailed information on some of the restrictions the TPD will impose. We will continue to update you as we obtain more clarity.
Maximum size of 10ml for e-liquid bottles
This is clearly going to be a major problem for all sorts of reasons, not least because it makes it a much more expensive and inconvenient way to purchase and store e-liquid since it’s customary for many vapers to use 5 or 10ml or even more, per day. It’s completely at odds with regulators’ desire to reduce waste and promote a green agenda in every other area of our lives.
Some commentators have indicated they suspect this size limit is so the products can be very easily taxed per bottle at a later date although the regulators state that this limit was set with user safety in mind, to reduce the possibility of serious effect should someone accidentally or purposefully drink a bottle of e-liquid. We find it strange that the same type of restriction has not been applied to other readily available products that could be equally or much more dangerous than e-liquid if ingested orally. No-one’s ever seen fit to limit purchases of bleach to a thimbleful or limit the maximum bottle size for alcoholic spirits to the equivalent of a couple of units worth. It’s even more nonsensical when you consider that this very same TPD legislation has done the exact opposite of this restriction for cigarettes, in banning their sale in packs of fewer than 20 cigarettes to make them less appealing and less affordable to children.
This maximum bottle size issue is further compounded by the fact that buy-one-get-one-free and some other types of price promotion offers will be banned although it’s not clear whether the restrictions on price promotions will be imposed immediately or at the end of the sell-through period3.
You will no longer be able to legally purchase nicotine containing liquid in bottles larger than 10ml in any EU member state once the sell-through period3 is over.
Maximum strength of 20mg/ml
Research studies4 have found that vaping is actually less effective at delivering nicotine than smoking, and vapers ingest less nicotine from vaping than smoking. Many indicators (and our own experience) tell us that a higher strength liquid is often needed by some users in their first attempts to make a complete switch away from smoking. We believe most of our customers who currently vape 24mg/ml will be able to reduce to 18mg/ml or 20mg/ml, but there does remain a core group of vapers who require strengths outside the typical range to keep them from going back to smoking and those vapers will need to reduce the strength they use and consider hardware changes that could make a lower strength liquid more satisfying. This restriction is again related to regulators’ misplaced concerns about accidental poisonings based on out-dated information about the lethal dose of nicotine due to dubious self-experiments in the nineteenth century5. There will be no such restriction placed on the nicotine level in products that are manufactured by pharmaceutical companies and approved under medicines legislation as a licensed medicinal product for smoking cessation, however, these will almost all be cigalike type devices with single-use/disposable high strength cartridges.
You will longer be able to purchase non-pharmaceutical e-liquid higher than 20mg/ml nicotine (whether it’s pre-mixed, ready-to-vape or high strength liquid for DIY mixing) legally in any EU member state once the sell-through period3 is over.
Maximum 2ml size for cartridges and tanks
According to the EU directive, most of the current advanced tanks will be banned outright. However, according to the UK draft legislation, it appears that this restriction may only apply to tanks that contain e-liquid at time of sale. It’s also not entirely clear as to whether this includes rebuildables or replacement heads and coils but the most recent communications from the UK government appear to indicate it includes anything which could contain nicotine in the form of e-liquid which is intended to be vaporised and inhaled. Most of the tanks we currently stock and similar ones in the near future may not be allowed under the new rules, and those few that do meet the restrictions may not be saleable unless they have been tested and a full dossier provided to the MHRA 6 months in advance of them being placed on sale3 (or by November 2016 if they are already on sale).
If you currently prefer tanks larger than 2ml, you may not be able to purchase them legally in any EU member state once the sell-through period3 is over. We await some further clarification of exactly how this restriction will be applied in the real world.
Leakproof re-filling mechanism
The regulators have not yet fully defined what they mean here, and there’s been some discussion about whether or not the regulators might require some sort of e-liquid bottle docking system – which would obviously mean consumers will be “locked-in” to different types of proprietary refilling mechanisms and thus limited to which brand of liquids could be used with certain brands of tanks. It is hoped that the definition will be more related to the size and length of the tip on bottles, and the filling hole size on tanks.
We await further information on how/what this requirement will mean in the real world.
Electronic cigarettes must provide a consistent nicotine dose
Every user has a different vaping style and so everyone’s “puff duration” and pressure differs. Added to that, modern devices allow you to tailor the experience to your preferences with most advanced devices currently available featuring adjustability in wattage or voltage, and airflow. Defining “normal use” is therefore very challenging and indeed no interpretation or specification has been given. It looks like each manufacturer will be required to formulate their own dosage testing protocol and demonstrate that the amount of nicotine delivered is consistent and repeatable using this standard protocol.
We await further information on what this requirement will mean in the real world.
Product testing and notification
UK Manufacturers and importers of electronic cigarettes and refill containers will be required to perform very comprehensive, costly testing and submit a notification to the Medicines and Healthcare Products Regulatory Agency (MHRA) 6 months prior to placing a new or substantially altered product on the market. For the UK, the notification will need to be presented in a specific way which meets the MHRA’s adopted format, and shall include:
- A list of all ingredients contained in the liquid and contained in the vapour produced.
- Toxicological data for all those ingredients within both the liquid and the vapour.
- Information on nicotine doses and uptake for the product.
- Full description of all components of the product including where applicable, the opening and refill mechanism of the product.
- A description of the production process and a declaration of conformity with the requirements of the directive.
- Annual sales volume reporting across the notified products including geographical information and preferences of various consumer groups.
There will obviously be huge financial implications involved in the testing and notification process, which has been based on a pharmaceutical model despite the fact that these products are not pharmaceutical or healthcare products, and indeed retailers will still not be allowed to present them as such, or make any claims as to their effectiveness as a quit-smoking method. It would have been sensible for regulations to require testing and identification of known potentially harmful inhalant risks, but the regulators have seen fit to demand the entire recipe for all liquids, thus forcing manufacturers to disclose proprietary formulations which are essentially, trade secrets.
Food flavouring manufacturers aren’t currently required to divulge their entire recipes and can retain some commercial confidentiality to prevent formulations being stolen or copied by competitors. This makes it extremely difficult to identify compounds present because chemical analysis is performed by looking for specific ingredients. Without having knowledge of what one’s looking for, analysis is almost impossible. Even where the compounds are identified by the flavouring manufacturer, e-liquid manufacturers will still need to run chemical analysis on every single iteration of their product (that’s every strength and flavour separately) to identify and quantify all the compounds present, both in the liquid, and in the vapour. Cost estimates vary wildly, depending on who you speak to, but a very conservative estimate is a minimum of £5000 per iteration (e.g. a banana flavour e-liquid in 4 strengths = a minimum of £20,000 just for the testing and documentation for that one flavour liquid). There are also the initial notification and annual renewal fees to factor in as well as the huge administrative burden.
The resulting impact of these provisions is not yet known because e-liquid is currently available in the UK in thousands of flavour and strength combinations, all of which will be unsaleable unless they have been tested and notified. Very few manufacturers outside the EU will comply with these reporting requirements, so it will be up to the retailer to undertake the testing and notification and bear the cost. This is unlikely to happen in many cases, so many non-EU products will simply disappear from the shelves. EU manufacturers and retailers who can’t afford to test and prepare notification for hundreds of different products will likely close their business, or radically cull their ranges.
Once manufacturers do take note and make provisions to adhere to the new testing and notification requirements, they will not be able to place products on sale until 6 months after they make their product notification submission to the MHRA.
Expect to see a massive reduction in the e-liquid choices available on the EU market once the sell-through period3 is over and a delay on new products coming to market. Also expect e-liquid prices to increase.
New packaging requirements
The TPD requires that e-cigarettes and e-liquid bottles contain the following information on the outside of their packaging:
- A list of all ingredients contained in the product in descending order of the weight including the nicotine content of the product.
- An indication of the nicotine delivery per dose.
- The batch number.
- A recommendation to keep the product out of reach of children.
- The warning “This product contains nicotine which is a highly addictive substance. It is not recommended for use by non-smokers”.
The box must also contain an enclosure leaflet with:
- Instructions for use and storage of the product, including a reference that the product is not recommended for use by young people and non-smokers.
- Contra-indications.
- Warnings for specific risk groups.
- Possible adverse effects; Addictiveness and toxicity.
- Contact details of the manufacturer or importer and a legal or natural contact person within the EU.
- The warning “This product contains nicotine which is a highly addictive substance. It is not recommended for use by non-smokers”.
A fold-out information leaflet attached to the bottle may be acceptable in place of a box with leaflet enclosure.
Technically, there isn’t a definition for what a “dose” is as it relates to vaping because we ALL vape completely differently. Every vaper decides for themselves what strength they use, and how much they vape so this requirement could be slightly challenging. There are concerns over possible liability issues where the product packaging contains contraindications, and warnings for specific groups (whilst omitting other specific groups) and indeed this requirement is going to be a challenging one for all non-pharmaceutical companies due to the limitations of general product liability insurance. Most current retailers don’t even have sufficient product liability insurance now due to its cost, and it’s highly unlikely any small-medium retailer could afford sufficient insurance for products where health warning information is contained on their products.
Along with the reduction in choice, you can also expect to see a much higher cost for e-liquid, that has been tested, notified, and packaged in accordance with the regulations. Other repercussions such as product liability insurance issues are unclear.
References
- http://ec.europa.eu/health/tobacco/docs/dir_201440_en.pdf
- The TPD definitions for “electronic cigarettes” and “refill containers”:
‘electronic cigarette’ means a product that can be used for consumption of nicotine-containing vapour via a mouth piece, or any component of that product, including a cartridge, a tank and the device without cartridge or tank. Electronic cigarettes can be disposable or refillable by means of a refill container and a tank, or rechargeable with single use cartridges;
‘refill container’ means a receptacle that contains a nicotine-containing liquid, which can be used to refill an electronic cigarette; - Deadlines
20 May 2016 - New regulations apply for new products brought onto the market after this date.
20 Nov 2016 - Deadline for manufacturers and importers to submit data on ingredients and emissions for existing products; and manufacturers and importers of e-cigarettes to notify existing products on the market (if they intend to continue selling them after May 2017).
20 May 2017 - Date by which sell-through period for existing stock of non-compliant products purchased before November 20th 2016 ends. - http://www.ecigarette-research.com/web/index.php/research/2014/155-ecig-nicotine
?http://www.nature.com/articles/srep04133 - How much Nicotine kills a Human? Tracing back the generally accepted lethal dose to dubious self-experiments in the nineteenth century –Bernd Mayer – Archives of Toxicology January 2014, Volume 88, Issue 1, pp5-7
http://link.springer.com/article/10.1007%2Fs00204-013-1127-0/fulltext.html
Other resources
Tobacco and Related Products Regulations 2016
The EU Tobacco Products Directive
The counterfactual (Clive Bates)
Write to your MP. It's too late to change the TPD, but the implementation and enforcement strategy is not yet set in stone.
Age Verification
On 1st October 2015, it became a criminal offence to supply nicotine containing e-cigs to persons under the age of 18.
We've always taken a firm position in this regard, however, as a result of this new legislation, we have implemented further steps to ensure sales to minors are prevented. These steps include requiring you to register for a customer account to shop on the site. We appreciate this may not feel as convenient as before, but we've decided it is the optimal solution to this new legislation.
Please click here for more information.
We've changed
We're thrilled to inform you that Cloud9Vaping will now be known as Crème de Vape. The great products, super-swift service and amazing customer support you've come to know and love, will remain - all wrapped up in a newer, classier package.
No need to change your login details or passwords, all your details have been brought across to the new site. The old Cloud9 web address will continue to work, but we'd recommend updating your bookmarks to www.cremedevape.com.

Liquid test results - update
The original content of the post, dated 28th June 2015 and entitled Liquid test results, was a table of test results of liquids supplied by Five Pawns (as well as other suppliers) and an explanation of those test results.
They declined to provide their own test results when we commenced trading at the start of 2015, accordingly (and in accordance with our product testing policy) we submitted 8 Five Pawns liquids to testing at an independent UKAS accredited laboratory.
We published the results approximately 7 weeks after providing them privately to Five Pawns.
Five Pawns took exception to the post, sent a cease and desist letter through their attorneys, threatening legal action if our post was not removed.
Whilst we stand by these results, we decided to take down the information to comply with Five Pawns’ request.
Our own solicitors have responded in robust terms rejecting allegations made by Five Pawns, and they have now released their own test results, and it is incontrovertible that liquid produced by Five Pawns contains the compounds in question.
Liquid test results
Edited: 29/06/15
The information relating to specific products withdrawn from sale as part of our testing/due diligence processes has been removed pending legal advice.
Update: 02/07/2015
Liquid test results - update
Update: 02/07/2015
Latest test results on our own brand liquid are now available.
Our liquid testing protocol
There's been a lot of discussion and debate about dangerous compounds that have been found in many flavoured e-liquids and food flavourings used in e-liquid production.
We felt it was about time to give a thorough explanation of exactly what we do, so that you can rest assured, we're taking every step we possibly can to provide as safe a product as possible, according to the current known risks.
We've been testing our products since 2011 at the same independant UK-based laboratory used by Trading Standards. The lab is accredited by the United Kingdom Accreditation service (UKAS) to the internationally recognised ISO 17025 standard which means they have demonstrated they are technically competant and have documented procedures and quality management systems in place to ensure they are producing precise, accurate and repeatable test data. This accreditation also means they are continually monitored and audited to ensure their procedures and methods continue to consistently provide accurate and valid results that can be trusted by governmental organisations and compared to test data produced by other accredited labs worldwide.
We don't generally publish our results alongside the liquids, because we anonymise the samples for company confidentiality. This "blind testing" policy is common in many industries, and was introduced to us by ECITA when we first began testing. This method wasn't an issue when we were simply running tests for our own internal due diligence. However, it has caused us to be unable to properly publish all of our own test results alongside the products on our site so we will be changing this policy in the future so we can publish all our test results with the product clearly identified.
Diethylene Glycol and Ethylene Glycol
Diethylene glycol (DEG) is a known contaminant that is often present in general reagent grade Glycerine that's not designed for ingestion or pharmaceutical use. Some research was released in 2009, claiming that the FDA sampled several pre-filled cartridges and found Diethylene Glycol (a poison) present. It's imperative that these compounds aren't contained in products designed for ingestion, although not much notice is given to this within our industry. We have historically tested all our liquids for both DEG and Ethylene Glycol to verify the purity of the diluents used in the product, and to-date, none of our pre-mixed e-liquids have ever tested positive for either of these compounds. We are proposing dropping this test from our protocol as we're confidentn our manufacturer's protocols eliminate all risks in this area.
Nicotine Assay
As nicotine can be dangerous in high doses, we feel it's prudent to test the actual nicotine level, in random e-liquid samples, both to confirm that it is within tolerance of the stated level, and to confirm it isn't present in liquids that are labelled zero nicotine. It has been our experience that some tobacco type flavour zero nicotine liquids can contain miniscule traces of nicotine, due to to the method of production involved in manufacturing some tobacco flavours, however, we have never seen any results above 0.04% nicotine (0.4mg/ml) in a zero nicotine e-liquid, and any product that tested above 0.02% (0.2mg/ml) would not be released for sale. The last time any of our zero liquids tested positive for traces of nicotine (at 0.01%) was in January 2013.
Propylene Glycol and Vegetable Glycerine Quantification
We run these tests primarily to ensure any product we advertise as "PG Free" is in fact completely free of PG, since these products will typically be chosen by consumers who have an allergy or an intolerance to Propylene Glycol. We also like to see the PG/VG ratios for our own consistency checks and so we can advise customers who have a particular PG/VG ratio preference.
Diketones
All flavourings used in our pre-mixed liquids, and available as concentrates for DIY mixing have been certified as safe for use in consumption via the digestive system - i.e. they are food-safe. However, this is not the whole story, since ingestion into the lungs via vapour is an entirely different pathway, and some flavourings that are completely safe to be eaten, are not safe to be vaped.
There are a family of compounds referred to as diketones, that pose some serious inhalation risks and should not be present in e-liquid. These compounds are used to provide a buttery/custard/vanilla type flavour and are currently found in many pre-mixed e- liquid ranges, particularly the more complex blends coming from the USA at the moment. The diketones of particular concern are Diacetyl (2,3-butanedione or DA) and 2,3-Pentanedione (Acetyl Propionyl or AP). We also test for the presence of Acetoin, which by itself, is not known to be harmful, but it can partially catalyse into DA under certain conditions, so we do test for acetoin, so that we are aware of the level, if present.
Diacetyl (DA) in its inhaled form, has been identified as the major cause of a serious lung condition observed amongst a group of popcorn factory workers exposed to large amounts of fumes containing DA in an industrial environment over an extended period of time. As yet, no tests have been performed on human inhalation via vaporisation of flavoured e-liquid, however, there is a published occupational safety limit of 66µg (66 parts per million), which means, if you vape 1ml of e-liquid, it must have less than 66µg or 0.0066% DA to be below the occupational safety limit. Our current testing is able to detect DA to 10 parts per million or 0.001%. None of our e-liquids or flavouings contain DA as a specific ingredient, and where we have identified it during testing, we have refused to stock the product or withdrawn it from sale if it was already on sale prior to receving DA-positive test results.
The dangers associated with inhaling DA have been known for some years, and we've been testing for the its presence since 2011, however, it was only in 2014 when Dr Farsalinos released the results of his research, that the industry realised flavouring manufacturers who were responding to the "noise" regarding Diacetyl, had been replacing it with Acetyl Propionyl (AP), and that AP has very similar inhalation risks and is not suitable for vaping either. Prior to this research, many vendors, including ourselves, were unaware that AP also posed a very real risk, and since this point, we have also included it in our testing protocol. The published occupational safety limit of AP is 135µg (135 parts per million), which means if you vape 1ml of e-liquid, it must have less than 135µg or 0.0135% AP in it to be below the occupational safety limit. Our current testing is able to detect AP to 10 parts per million or 0.001%.
As of the date of this article, we do not stock any pre-mixed e-liquids that have tested positive for DA or AP at any level.
We may take the view in future, that some consumers want these types of flavours, and we may decide to stock e-liquid that has tested positive for DA at a very low level, as it occurs naturally in some flavours. If we did this, we would impose a limit ourselves, of approximately 25 parts per million (0.0025%). If we stock any e-liquids in future that do contain traces of DA up to 25 parts per million (0.0025%) we will clearly indicate this information. Where our testing identifies an e-liquid contains DA above 25 parts per million (0.0025%), we will still continue to refuse to stock it. We are unlikely to alter our position on AP, since it never occurs naturally and is only present when it has been physically added.
Flavour Concentrates
We do also use the same testing protocol (excluding nicotine assay) on specific flavour concentrates where we believe, by virtue of their flavour profile, could contain harmful diketones. None of our concentrates have ever tested positive for Diacetyl, but some do contain AP in very low quantities. Where concentrated flavourings are concerned, assuming the concentrate is used at 10% and 5ml per day is vaped, our own self-imposed limit would translate to 250 parts per million (0.025%). We will not stock any new flavour concentrates that exceed this level, and those we currently stock that could exceed this level are clearly identified in our flavour concentrates product information download.
Further information
It should be noted that where a liquid has a buttery or custardy taste, but has tested free of DA or AP, the buttery note will have been achieved by Acetoin or Butyric acid, or a combination of these components. Butyric acid can cause irritation in some vapers and it can also be responsible for an unpleasant sour after-taste in certain concentrations or flavour combinations.
The amount of liquid you vape per day may exceed averages, thus exposing you to more risk, and indeed, if you are at all concerned about diketones, or other as-yet unidentified, potentially harmful component, we advise you steer clear of all buttery, custardy, vanilla, creamy & sweet flavours or consider vaping subtly flavoured, or even unflavoured liquids, rather than strongly flavoured recipes with many flavouring components present, in high concentrations.
Additionally, it should be remembered that vaping is a "reduced-harm" alternative to smoking - this doesn't mean it's completely harmless but that it's known to be much less harmful than smoking traditional combustible tobacco. The fact that liquids have tested free of these known risks, is not an indication that any one flavouring is more or less "safe" than any other - no long term or comprehensive studies have yet been performed on human inhalation of vaporised food flavourings.
Positive Action
In the recent past, we removed a well-known premium e-liquid range from sale because our test results indicated very high levels of DA (testing showed up to 100 ppm or 0.01%), and extremely high levels of AP (testing showed up to 2500 ppm or 0.25%). We trusted the manufacturer were doing their own due diligence but this experience was a reminder that there are no shortcuts. We are not prepared to take risks with your health and our company reputation, and will not stock any new liquid products until the supplier has provided comprehensive analysis performed by a known, accredited & independent laboratory, or we have performed our own analysis.
Updating the YiHi SX Mini M Class
One of the great features of the YiHi SX Mini M-Class is that its firmware can be upgraded by the owner.
In this video, Phil Busardo takes you through the process of updating the YiHi SX Mini M-Class to the latest firmware.
2014 UKVapers.org Awards
We're extremely proud to have been nominated and to have come top in three categories again in the 2014 UK Vapers awards. Winning these awards 2 years running is an amazing achievement and we'd like to extend enormous thanks to all those who voted and to all our customers for making 2014 another amazing, award winning year!
Best UK Based vendor - Cloud 9 Vaping
Best Customer Service - Cloud 9 Vaping
Best “High end” vendor - Cloud 9 Vaping
Some of our products also came out on top:
Eleaf iStick - Best budget mod
EVOLV DNA40 - Vaping Innovation 2014
Aspire Nautilus and Aspire Nautilus Mini - Best budget tank atomiser
Note: We were formerly known as Cloud9Vaping and rebranded as Creme de Vape in 2015.
Charger Safety
Electrical Safety First have asked us to support their campaign to raise awareness about the dangers of purchasing cheap and untested e-cig equipment.
We take the subject of electrical safety very seriously, and it's always worth reminding vapers to buy high quality branded equipment from reputable retailers and learn how use it safely.
Please visit the Electrical Safety First website for more information.
2014 Ecig Click Awards
We're extremely proud to have been nominated and to have come top in 3 categories again in the 2014 EcigClick awards.
Best Overall - Cloud 9 Vaping
Best Customer Service - Cloud 9 Vaping
Best One Stop Shop - Cloud 9 Vaping
Some of our products also came out on top:
SvoeMesto Kayfun Lite/V3.1 - Best Rebuildable atomiser/tank
Eleaf iStick - Best Box Mod
Aspire Nautilus Mini - Best Clearomiser/tank
Innokin MVP2 - Best Advanced Personal Vaporiser
Enormous thanks to all who voted and to all our customers for making 2014 another award winning year.
Note: We were formerly known as Cloud9Vaping and rebranded as Creme de Vape in 2015.
Counterfeiting
Non-authorised copies and counterfeits are a major problem across all product sectors, but have really got out of control in recent months within the electronic cigarette industry. As a fairly young industry, with many unique brands entering the market each week it’s a very difficult problem to solve, although we have already begun to hear stories of original manufacturers fighting back hard, getting shipments seized and destroyed at the border and getting the PayPal accounts of the dealers of these illegal goods frozen. We can only hope this will become a more common occurrence.
Counterfeits and clones, what’s the difference?
A counterfeit product has every element of the product copied, including the original maker’s marks and logos, or can be a slightly or entirely differently designed product, featuring a known manufacturer’s logo or marks so as to make it appear as if it is made by the manufacturer who owns the logo. Often, the consumer will not have any idea they are purchasing a counterfeit product, and sometimes, even the retailer will have been duped by their supplier too.
Cloning is where most elements of the product have been copied, but the product is intended to be identified and sold as a copy, and doesn’t feature the logo or marks of the original manufacturer. Although it has very serious effects on the designer/manufacturer of the original product, as well as its entire distribution network, it’s harder to prevent because the process of proving intellectual property of designs is harder and very costly, even when a patent is owned.
Essentially, whether you call them counterfeits, copies or clones, they are all theft of original work and made using the intellectual effort and resources of the original innovators.
A bargain, or is it?
You might not regard buying a counterfeit mod or atomiser as an illegal transaction, but merely a cheaper way to get the latest device everyone’s talking about. Often the temptation for consumers can be too strong to resist, with many not understanding the very real risks and ramifications attached to this illicit industry.
Besides the very obvious quality issues usually found on counterfeit and clone products, which at their very least, often lead to user experience issues because of poor performance or defective operation, or their visual appearance or “finish” being below par, there is a much more worrying safety concern with these fake products. The safety of a product can be impacted by means of dangerous materials finding their way into the finished products (for example, high levels of lead in cheaper metal alloys) or worse, major electrical faults which could lead to very dangerous explosions and fires despite the user ensuring they observe all the normal safety measures.
I can’t afford genuine products, what’s the harm in buying a clone?
Many consumers, particularly in our market, seem very happy to knowingly purchase knock-off goods. Maybe this is because of limited funds, or simply because they can’t justify paying the full price for the equipment they so desperately want to acquire. There is an element that even believes the original product is “so expensive that it deserves to be cloned”.
If you are one of those that think like this, I ask you to stop right there and think a little more deeply about the costs. Imagine for a moment that you spent time and money coming up with a great piece of work, or a product, would you be happy to have invested hours and hours of your time, perhaps time you’d have rather spent with your family, and borrowed or invested thousands of pounds in your idea, only to have someone come along as soon as you release it, and copy it for their own gain and thereby halt any hope of you recouping your original costs?
If you believe you yourself deserve to be rewarded properly for your own talents and effort, and you appreciate the talent and the work of one of the many wonderful and talented manufacturers in our industry, surely you believe they should also be rewarded properly for their work, rather than the efforts and toil being “used” by someone else for material gain?
Consumers also need to be aware that the type of companies engaging in counterfeiting practices will have no concern about the safety of the products, or the safety of their manufacturing processes, and are often engaged in organised crime and/or using child labour to keep their costs as low as possible.
The Impact of Clones and Counterfeits
Counterfeit products lead directly to financial turmoil for businesses such as low turnover, stolen know-how, lost jobs, lawsuits and price hikes, followed by growth halt & innovation slowing. The original manufacturer relies on the sales of their original product to pay them back for all their time and investment in the original design, prototype production, manufacturing costs and testing phases involved in bringing a new product to market, as well as requiring the funds to help toward future research and innovation.
The inevitable drop in sales of the legitimate product means these original manufacturing costs may never be fully recovered, or that limited funds remain for further research, innovation and development.
Authorised retailers of a product which has been cloned or faked may find themselves completely unable to sell the original products they have on hand, or only able to sell them at a loss. Without going into great detail here about stock management or the cash conversion cycle, having funds tied-up in un-saleable stock is a massive drain on a businesses’ resources, both in terms of the funds not being available to pay employees or purchase other stock to maintain profitability, and also in terms of the space and resources used by such product whilst it remains sitting in the warehouse.
Also consider the negative impression a poorly performing counterfeit product leads to in the mind of the consumer, who may have no idea it’s not a genuine product. As the saying goes, “mud sticks” and the negative experience will forever be associated with that of the original manufacturer in the mind of the consumer.
The ramifications of counterfeiting aren’t just limited to those involved in the trade – it actually affects everyone in an economy affected by the activity, with Governments, businesses and society being robbed of tax revenue, business income and jobs. The flood of counterfeit products leads to more public spending through the requirements for increased border efforts to spot, and seize the fakes as they are imported, increased Trading Standards’ resources and other enforcement work to counter the illicit trade. It also increases the burden on the government caused by unemployment and low pay (analysis has been performed which indicates 300,000 jobs have been lost in the EU wholly as a result of counterfeiting activity); and it increases the price of legitimate products as companies seek to recoup their losses.
The effort of dealing with the problem, both in financial terms and also in the emotional toll it takes on original manufacturers and their authorised retailers, could mean that they simply lose hope for the industry they love being a part of, and give up and go and do something else instead.
Consumer power
There is a great need, particularly within our industry, to build more awareness of the scale and effects of the problem. Border controls, Trading Standards, merchant payment processors, sales sites (such as eBay and Amazon), Official Trade Bodies and forum owners all need to take a much tougher stand against counterfeiters, and although the responsibility falls on everyone, the greatest change can be brought about by consumers themselves who can to exert their influence with their purchasing choices.
While consumers often cannot identify counterfeit goods, the more obvious routes of supply should be shunned and companies engaging in this trade should be named and shamed. You can avoid contributing to this problem by only going to well-known and respected authorised dealers, and if you are in any doubt, try to contact the original manufacturer and ask them if the supplier you’re about to purchase from is an authorised one. It’s understood that you may find fakes at your local market, but also be aware that they are often sold on professional looking websites.
Stay alert to warning signs. If the product is being sold by someone other than an authorised retailer, steer clear. If the price is too good to be true, then it probably is.
Conclusion
The bottom line is that counterfeiting costs everyone, and those too concerned with paying the cheapest price for their vaping kit, when they should be focused on the harmful effects that these cheaply made counterfeits could have, are the primary drivers for the rampant counterfeiting within our industry. If people refuse to purchase knock-off products, these rogue industries will suffer economically and ultimately disappear.
Vapefest 2014
Vapefest 2014 promises to be bigger and better than ever before.
As ever, it's a fabulous opportunity to meet up with fellow vapers and catch up with old friends.
We'll be donating some wonderful raffle prizes, so do be sure to enter, and don't forget to let us know if you would like us to bring anything in particular with us.
See you there!